Plant Signal Behav. 2013 Sep 1; 8(9): e25504.
Gibberellic acid in plant
Abstract
Gibberellic acid (GA), a plant hormone stimulating plant growth and development, is a tetracyclic di-terpenoid compound. GAs stimulate seed germination, trigger transitions from meristem to shoot growth, juvenile to adult leaf stage, vegetative to flowering, determines sex expression and grain development along with an interaction of different environmental factors viz., light, temperature and water. The major site of bioactive GA is stamens that influence male flower production and pedicel growth. However, this opens up the question of how female flowers regulate growth and development, since regulatory mechanisms/organs other than those in male flowers are mandatory. Although GAs are thought to act occasionally like paracrine signals do, it is still a mystery to understand the GA biosynthesis and its movement. It has not yet confirmed the appropriate site of bioactive GA in plants or which tissues targeted by bioactive GAs to initiate their action. Presently, it is a great challenge for scientific community to understand the appropriate mechanism of GA movement in plant’s growth, floral development, sex expression, grain development and seed germination. The appropriate elucidation of GA transport mechanism is essential for the survival of plant species and successful crop production.
History and Evolution
Gibberellins commonly known as gibberellic acids first came to the attention of western scientists in 1950s, they had been discovered much earlier in Japan. Rice farmers of Japan had long known of a fungal disease called foolish seedling or bakanae disease in Japanese that causes rice plants to grow taller and eliminated seed production. Plant pathologists found that these symptoms in rice plant were induced by a chemical secreted by a pathogenic fungus, Gibberella fujikuroi. Culturing this fungus in the laboratory and analyzing the culture filtrate enabled Japanese scientists in the 1930s to obtain impure crystal of two fungal “compounds” possessing plant growth promoting activity. One of these, because it was isolated from the fungus Gibberella, was named gibberellin A. In 1950s scientists of Tokyo University separated and characterized 3 different gibberellins from gibberellin A sample, and named them gibberellin A1, gibberellin A2 and gibberellin A3. The numbering system for gibberellins used in the past 50 y builds on this initial nomenclature of gibberellins A1 (GA1), GA2, and GA3.
In the same year, 2 research groups, one at Imperial Chemical Industries in Britain and other at the US Department of Agriculture (USDA) in Illinois, elucidated the chemical structure of the compound that they had purified from Gibberella culture filtration and named gibberellic acid. This compound was later shown to be identical to the gibberellin isolated by the Japanese scientist. For this GA3 is also referred to as gibberellic acid. GA3 is the principal component in Gibberella culture. The GA3 is the most frequently produced GA in commercial industrial scale fermentations of Gibberella for agronomic, horticultural and other scientific uses. Identification of a GA from a plant extract was first made in 1958 with the discovery of GA1 from immature seeds of vuner bean (Phaseolus cocineus). As more and more GAs from Gibberella and different plant sources were characterized, a scheme was adopted in 1968 to number them (GA1–GA4), in chronological order of their discovery.
Gibberellin Biosynthesis
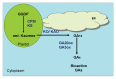
Gibberellins (GAs) are endogenous plant growth regulators, having tetracyclic, diterpenoid compounds. After valuable efforts to understand the GA biosynthesis and movements, the appropriate site of bioactive GA in plants or tissues targeted by bioactive GAs to initiate their action has not yet been confirmed. Dwarf plant bioassay and its quantitative analysis revealed the presence of GA in active growing tissues i.e., shoot apices, young leaves and flowers. In contrast, there are some reports for the presence of GAs in xylem and phloem exudates, indicating a long-distance transport of Gas.4,5 The transport of active GAs and their intermediates was supported by grafting experiments. The contradictory results obtained from different experiments could not pin-point the site of synthesis of bioactive GA. Gibberellins being synthesized via the terpenoid pathway, require 3 enzymes viz., terpene synthase (TPSs), cytochrome P450 monooxygenase (P450s) and 2-oxoglutarate dependent dehydrogenase (2 ODDs), for the biosynthesis of bioactive GA from GGDP in plants (Fig. 1). Two terpene synthase, ent-copalyl diphosphate synthase (CPS) and ent-kaurene synthase (KS), located in plastids, involved in conversion of GGDP to tetracyclic hydrocarbon intermediate ent-kaurene (Fig. 1). ent-Kaurene is then converted to GA12 by 2 P450s. First, ent-Kaurene oxidase (KO) present in the outer membrane of plastid, catalyzes the sequential oxidation on C-19 to produce ent-kaurenoic acid. Second, ent kaurenoic acid oxidase (KAO) present in endoplasmic reticulum is subsequently converted to GA12. Bioactive GA4 is converted from GA12 through oxidations on C-20 and C-3 by GA 20-oxidase (GA20ox) and GA 3-oxidase (GA3ox), respectively (Fig. 1).
GA Signaling in Dormancy and Seed Germination
Seed contains embryo that is arrested to develop in to plant with appropriate environmental conditions to continue their life cycles. Breaking of seed dormancy to germination is controlled by some physical factors (light, temperature and moisture) and by the endogenous growth regulating hormones (GA and ABA). GA stimulates the seed germination whereas, ABA is involved in the establishment and maintenance of dormancy. GA exerts its influence in two manners, first by increasing the growth potential of embryo and second by inducing hydrolytic enzymes. During seed germination embryonic GA is released that triggers the weakness of seed cover by stimulating gene expression involved in cell expansion and modification as reported in Arabidopsis. GAs represent a natural regulator of the processes involved in seed germination to stimulate the production of hydrolytic enzyme i.e., ά-amylase, in the aleuron layer of germinating cereal grains. Cereal grains can be divided into 3 parts i.e., embryo, endosperm and seed coat. The endosperm is composed of the aleuron layer and centrally located starchy endosperm. The starchy endosperm, typically non-living at maturity, consists of thin walled cells with starch grains surrounded by aleuron layer, having thick cell wall with protein bodies. As consequences, the stored food reserves of the starchy endosperms are broken down into soluble sugars, amino acids, and other products that are transported to the growing embryo. GA biosynthetic enzymes, GA 20-oxidase and GA 3-oxidase genes show tissue and cell specific patterns of expression in germinating grain of rice, although this expression is confined to the epithelium and developing short tissues of germinating embryo. The embryo seems to be a site of GA biosynthesis and response, whereas aleurone layer shows site of response only. The response is not same in both the locations. In the aleurone, synthesis of ά-amylose takes place, whereas in developing shoot cell division/elongation. The expression of α-amylase gene is upregulated by exogenous GA, mediated through SLN1 and GAMYB transcription factors. On the other hand, PKABA1, an ABA-responsive serine/threonine protein kinase, inhibits gene expression in barley. GA not only restricts to the secretion of hydrolytic enzymes but, trigger the programmed cell death, combining with reactive oxygen species. The aleuron gene expression pattern has identified many new genes whose regulation is up/downregulated by GA and ABA treatment in barley. Mutation in a gene encoding a heterotrimeric GA protein impairs GA signaling in aleurone cells causes dwarf phenotype in rice. Radical emergence requires breaking the endosperm caps, a major physical restraint to germination in tomato and tobacco. The GA-deficient-1 (gib-1) mutant of tomato and Arabidopsis ga1–3 mutant could not germinate without exogenous GA application, however it germinated when endosperm caps were removed. GA plays an important role in the endosperm cap weakening. The bioactive GAs are produced in embryo and transported to aleurone layer, 27and trigger the expression of ά-amylase was confirmed after physiological and biochemical characterization. It is concluded that during seed germination the aleurone layer is unable to synthesize GA but perceive the GA signals.
GA Biosynthesis and Signaling in the Apical Meristem
Physiological studies and phenotypic characterization of mutants with impaired GA biosynthesis revealed that GA plays an important role in internode elongation. It stimulates cell division and expansion in response to light or dark (photomorphogenesis and skotomorphogenesis). Despite complexity, the GA biosynthetic pathway has been well characterized. It is very difficult to determine precisely the site of bioactive GA biosynthesis in plants. Very little is known about level of GA in plants and still much remain to understand the signal transduction pathways leading to elongation of stems and leaves with response to different environmental factors. Various studies on gene expression and characterization of GA deficient mutants revealed GA signaling and bioactive sites in plants. A model proposed by Sakamoto depicted relationship between GA biosynthesis and cell fate determination at the apical region of tobacco shoot. A KNOTTED1-like homeobox (KNOX) protein, NTH15 is present at the corpus region of the shoot apical meristem (SAM). An interaction with the cis-acting element results a negative regulation of the GA 20-oxidase gene. When NTH15 expression is controlled, GA biosynthesis starts and finally stimulates cell division and determines cell fate. In rice, the expression of GA related genes is restricted to the basal and peripheral region of the SAM rather than corpus region. In rice corpus region of SAM expressed OSHI and KNOX type homeobox genes to determine cell fate. Another report also revealed the expression of GA regulated genes in growing tisuues of Arabidopsis. GA promotes cell elongation through releasing DELLA mediated inhibition of BZR1 transcription factor.
GA in the Flowering and Sex Expression
GAs regulates flower initiation and its development and it is essential for male and female fertility not for differentiation of floral organs. GA-deficient mutants in Arabidopsis and tomato showed abnormal stamen development, while extreme GA deficiency revealed female sterility. No viable pollen develops in severe GA-deficient mutants, and sepals, petals, and pistils are underdeveloped, leading in some cases even to premature abortion of the flower. Application of bioactive GAs or even of the GA precursor GA9 restores normal flower development. Arabidopsis stamens require higher GA concentration than do the other floral organs stamens offer a rich source for GAs, as has been demonstrated in rice. Moreover, for a long time it has been known that in Glechoma hederacea, stamen-derived GAs stimulate corolla growth. Griffiths found that not only the stamen and petal development and arrested and the pistil length reduced, but also reduced the pedical elongation in triple GID1 receptor mutants of Arabidopsis. Further, Hu et al. identified stamens and/or flower receptacles as 2 potential sites for bioactive GA synthesis in Arabidopsis flowers, and suggest that GAs are transported from these organs to promote petal growth. GA-deficit mutants produced short stamen, resulting shortening in filaments and compromised self-pollination. The tapetum, essential for pollen development providing nutrients, contains pollen coat and allowing dehiscence. The tapetum seems to be a major site of GA biosynthesis in developing anthers in rice and Arabidopsis. The expression of GA genes was reported in anthers only after meiosis45 and it is interesting to speculate on the extent and distance GAs are exported from anthers. GA plays very important role in pollen germination and pollen tube growth. Pollens in GA deficit mutants do not germinate unless rescued by exogenous GA. Late stamen development (filament elongation, anther dehiscence, and pollen maturation) regulated through GA in coordination with jasmonic acid, whereas the GA alone mediated early anther development.
GA in pollen itself increases (7-fold) during pollen tube growth, but this may be species specific. Pollen is a rich source of GA and its content may be 200 fold greater than that in the ovary tissue both in Petunia hybrida and Lillium. However, this level of pollen GA contributes little to total ovary GA at the time of pollination. Within hour of its germination, pollens’ GA activity decrease drastically in Petunia and Lilium. Later in germination pollen tube growth becomes slow, and this might be reflected in decrease in bioactive GAs at this time, especially in angiosperm pollen. However, too little is known about differences in the timing of these changes during pollen tube growth on GA conversion, and in native GA type. Gibberellin is also reported to control sex expression by plant growth regulators. In cucumber GA3 treatments promote the male tendency in both gyonecious and hermaphroditic lines. Self-pollination study of female cucumber lines responded to repeated GA3 treatment to such an extent that the continuous female phase could be prevented. There are indication that GA do not directly promote stamen differentiation in the embryonal floral bud but merely suppress female flower formation and that, in the lack of the latter, male flower ultimately develop. However, in bitter gourd GA3 at lower concentration promoted induction of female flowers and improved the fruit quality. The highest number of female flowers per vine was recorded in bitter gourd with 50 ppm GA3. It also stimulates the pistillate flower development in castor bean, corn and hyoscyamus.
GA in Embryo Development
Gibberellins (GAs) are important constituent to regulate the temporal organization of maturation phase in maize. Early embryogenesis in maize accumulated more bioactive GAs and the concentration decline as ABA level rises. Similar relationship between GA and ABA was reported in barley and wheat. GA and ABA being antagonistic to each other maintain the relation between vivpary and quiescence; occur at or before stage 2 of embryo development. However, the level at which gene expression is affected by GA modulation remains to be determined.
Conclusion
Seed germination, stem elongation, meristmetic tissue development and differentiation of floral organs are highly dependent on GA signaling system and mechanism. GA is required to break seed dormancy leading to its germination. Seed germination is a complex process, controlled by both physical and internal regulating factors. GA plays very important role in controlling and promoting germination in cereal grains and other crop species. It is confirmed that GA deficit mutants failed to germinate in absence of exogenous GA. However, a very small known GA signaling factors has been shown to mediate the regulation of seed germination. Physiological studies and phenotypic characterization of mutants impaired GA biosynthesis. It revealed that GA plays an important role in stem or internode elongation. It stimulates cell division and expansion in response to light or dark. GAs regulate flower initiation in some LD and biennial species and inhibit flowering of some perennials, and its development and it is essential for male and female fertility but not for the specification and differentiation of floral organs. GA3 treatment promotes the male tendency in both gyonecious and hermaphroditic lines in some species. Three major points that are involved in the GA signaling mechanism are 1) the stamen is the essential site of GA synthesis, other sites cannot replace the stamen; 2) GA20ox and GA3ox are key regulators of GA biosynthesis in the stamen and 3) short-distance movement of bioactive GA (but not of its biosynthetic precursors) from the stamen to the other floral organs and the pedicel is essential and sufficient for flower development. Thus, the stamen is the site that regulates, via bioactive GA, the male flower and the pedicel growth. However, this opens up the question of how female flowers regulate growth and development, since regulatory mechanisms/organs other than those in male flowers are mandatory. Although GAs are thought to act occasionally like paracrine signals do, but it is still a mystery to understand the exact mechanism of gibberellic acid movement/transport in plants. Presently, it is a challenge for scientific community to understand the appropriate molecular mechanism of GA movement in plant’s cell. It is still a mystery to understand the exact mechanism of gibberellic acid in plant growth, floral development, sex expression, grain development and seed germination. The appropriate elucidation of GA transport mechanism is essential for the survival of plant species and successful crop production.
Footnotes
Previously published online: www.landesbioscience.com/journals/psb/article/25504